yUcK!
RE Israeli "booster success"? UK's infinite wave and silent boosters, Molnupiravir assumptions, etc.
A topical post, to cap off the week. On the docket:
Forever Spike This!
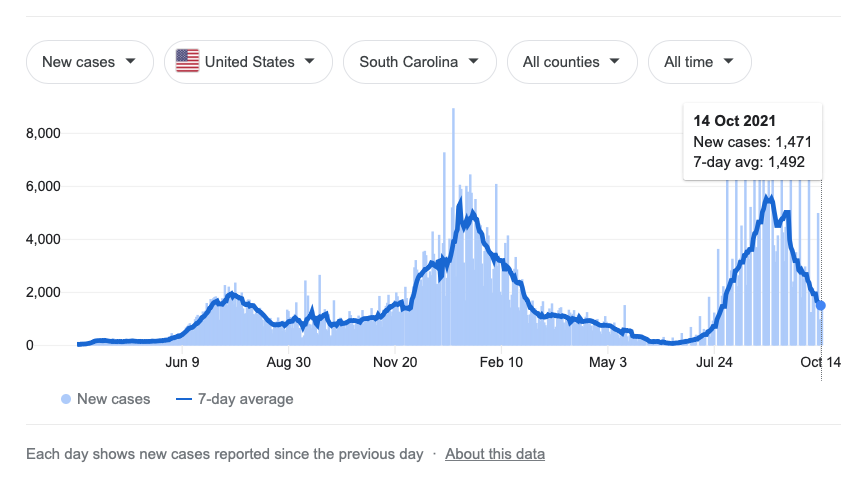
Cases have plummeted in Israel, happily defying the catastrophic fears I voiced in Forever Spike - namely that “waves” would be replaced by “plateaus” everywhere the Covid vaccine had been rolled out:
The reader is invited to formulate their own bet for whether this is a byproduct of the booster campaign launched at the end of July, or of the more synchronous roll-out of the vaccine among the primary eligible age groups in Israel - or of both, or neither.
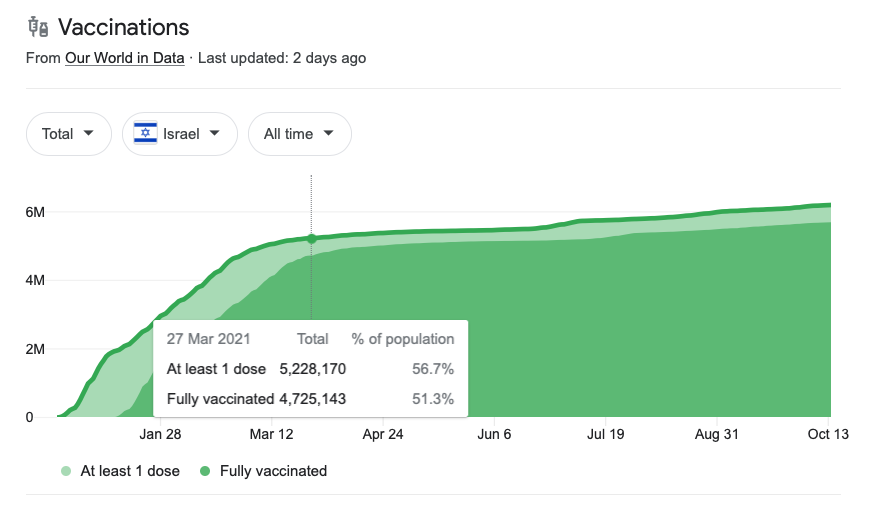
Regarding the second point, Israel’s vaccine roll-out is sui generis among larger countries in its rapid climb to the eventual resting point:
Since almost all the Covid-vaccinated finished leaving the ~4 month “infection efficacy” window in July, there was no longer a constant input of susceptible 4+ month-vaccinated afterward. And with no more constant input of fresh kindling, the wave burnt out.
Regarding the last of those suggestions, neither the booster roll-out or the “done in March” explanations apply to the southern US, where the summer wave is also retreating with a “natural” temperament. Here is Floridamanland:
Florida’s vaccine uptake rate is almost a perfect case-study for Forever Spike - there should not be a single week for the rest of the year when ~1.5% of Floridians are not exiting the ~4 month window of “infection efficacy” protection. And yet, no plateau. “Virus gonna virus” remains an immortal truth.
The patterns in the southern US thus suggest Israel could have done things quite differently without any change to the summer wave. However, there is at least a bit of ambiguity in some southern states,2 which suggests things might get wobbly again - while in Israel and Florida the summer waves appear ripe for their respective “time of death” declarations.
The suggestion in my “Forever Spike Disaster!” scenario, that the innate immunity of the unvaccinated would be subjected to prolonged, wave-like stress-testing, with potential deleterious effects, can hopefully be set aside. Unless you’re in the UK…
yUcK!
(For an update notice on this segment, see Footnote 5.)
It seemed certain, a month ago, that the UK was merely an early adopter showing the rest of the West what we were in for. But now that waves have reversed in so many other regions, the UK has become not a frontrunner but an aberration. What exactly is going on over there?
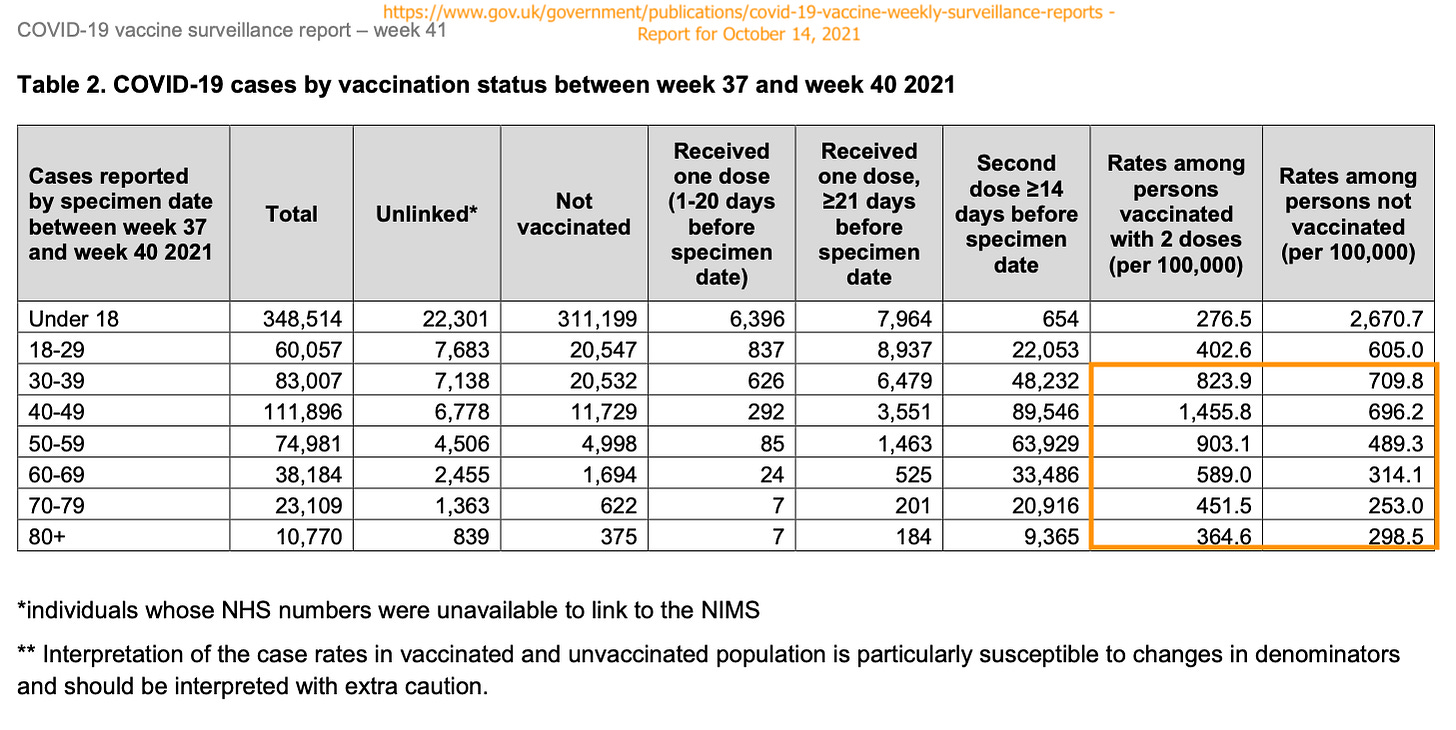
Again, at this point I will only suggest candidates for the reader’s own personal guess. The signal for “negative infection efficacy” rolling about ~4 months after vaccination uptake remains quite legible. In September, the Covid-vaccinated over 40 in England were scoring positive for “cases” at a higher per-cap rate in real-time (measuring only the preceding three weeks) than the unvaccinated. Now, that holds true for the over 30 cohort as well. In yesterday’s update to the UK Health Security Agency report:4
If all “unlinked” cases were counted as unvaccinated, the 30-39 unvaccinated positivity rate would go a bit higher than for the 2-dose vaccinated; the 40-49 unvaccinated positivity rate would still be lower. So, it’s possible that the 30-39 Covid-vaccinated group are still in a more transitional level between protection and detriment. (Additionally, I have taken the overall increase in case rates everywhere the Covid vaccines have been rolled out as a strong argument that real-time “negative infection efficacy” is not merely an artifact of the Covid vaccinated catching up with the unvaccinated. Additionally, as before, “severe outcome efficacy” remains quite robust.5) This “transitional” state matches the timing of Covid vaccine uptake for 30-39 year-olds:
Half of the Covid-vaccinated 30-39 year-olds are over 4 months post-vaccine, half are under. But, again, we can take the situation in the southern US as a strong argument to throw this theory out the window.
Additionally, the Covid-vaccinated can no longer be considered the major drivers in cases in the latest “K” leg. Rather, it is the young who have returned to school. Comparing 3-week cases by age group in the chart above, almost half of confirmed cases are in the Under 18 cohort:
348514/(348514+60057+83007+111896+74981+38184+23109+10770) = .464
The Agency’s “Flu and Covid surveillance” update on the same day confirms that rates are increasing more for (mostly unvaccinated) children than for the mixed bag of young adults:6
It seems plausible that school is not really increasing cases among the young compared to other groups, but “cases” - the 5 to 9 cohort in particular are probably being tested and confirmed positive at a higher rate than in the summer, despite presumably fewer symptoms. Without this excessive testing, the current “K” leg might be more of a plateau (which still leaves the UK as an outlier in wave persistence).
One more thing to consider is boosters, which were approved in the UK without much fanfare last month, though with the rollout preceding gradually by age, and much more narrow exceptions for those under 50 than in the US.7 So far, there have only been 2 million booster doses recorded.8 Still, this could be having all sorts of distorting effects on the “persons vaccinated with 2 doses” group in the latest report, and going forward.
In general, the last reliable window into “2 dose only” efficacy is being plastered over. History will never really know what a world without “boosting” would have looked like.
Yuck!
Oh... Canada.
Meanwhile, the northeastern US doesn’t seem destined to replay the summer wave scripts of either the UK or the south. The “autumn wave” seems to have already reversed in some northeastern states, without ever reaching the heights seen elsewhere in the summer. Massachusetts is a typical case:
We can first consider boosters, again, as a possible factor in reversing the wave by removing individuals from the vulnerable population. Current reports do not suggest anything like a massive embrace of triple-dosing, despite polling from The Asylum suggesting widespread popularity among the already Covid-vaccinated.10 These reports, however, may not account for widespread “booster fraud” - especially if individuals who originally opted for the Moderna or Janssen pseudo-vaccines did not want to wait for the corresponding FDA blessing.
Or, the reader can ascribe this happy outcome to the high moral character of the citizens of the northeast, who understand the importance of conforming to whatever it is unelected dictators tell them to do at a given moment.
But an additional factor I would like to propose is the spring wave. Most states in the northeast, Great Lakes region, and Washington and Oregon had legible waves in March and April. In fact this extends all the way down the east coast, and is just-visible in Florida, which makes sense if you are familiar with the gravitational power of the I-95. The association of the east coast, Great Lakes, and Pacific northwest with a spring wave suggest an obvious relationship with the giant wave in Canada around that same time:11
Thus, these regions seem to have been (mostly12) spared the embarrassment heaped onto the south this summer by burning off some extra fuel in the spring. New York, however, demonstrates that the spring wavers might not be heading for such rosy outcomes after all. The UK, it turns out, may soon have more company in Forever Spike Purgatory:
The failure of ribonucleoside/ribonucleotide analogs during clinical development has been due to toxicity that was not detected during preclinical studies.14
Molnupiravir Myths?!
An interview at MedPage Today caught my eye this morning, partly for the subject seeming to have no idea how Molnupiravir works and partly for seeming to know likely realities about the drug’s development and pending approval that are not apparent to an outsider. This consisted of the transcript of the “Track the Vax” podcast, which discussed the pill with Vanderbilt University professor William Schaffner:15
Schaffner: It's kind of similar to Tamiflu, which we give for influenza infection. And so we hope that this will get approved and can be used more widely.
Molnupiravir is not “kind of similar to Tamiflu.” As discussed in “Doppelgänger,” it is “kind of similar” to antivirals which are known to cause birth defects in every animal ever studied:
Teratogenic and/or embryocidal effects demonstrated in all animal species exposed to Ribavirin [another nucleoside analog marketed as an “antiviral”]. Ribavirin has a long half-life (12 days after multiple doses [and this does not account for intracellular residues]) and may persist in nonplasma compartments for as long as 6 months.16
Tamiflu is merely a “neuraminidase inhibitor,” which interferes with the mechanical function of the protein influenza needs to unstick from host cells, though that doesn’t mean it is without possible harms. But apparently I am a better expert on biology than Vanderbilt infectious disease professors at this point.
What is interesting about the transcript, nonetheless, are claims made regarding the drug’s development and approval. Schaffner portrays the FDA as still having agency in the review and pending approval, suggesting that things hinge largely on the data - whereas, to an outsider, it seems as if there is more or less an unoccupied room somewhere with a candle burning a string that is holding up a rubber stamp.
More to the point, Schaffner claims to be colleagues with one of Molnupiravir’s initial developers. He asserts, based on this peripheral familiarity with the process, that the drug has already been aggressively screened for ability to cause mutations in the host cell. In the most significant exchange:
Marshall: So what do we know about those possible side effects?
Schaffner: Actually, the side effect profile has not been widely released yet. And I haven't seen that. That's going to be of great interest to the Food and Drug Administration and to its advisory committee. So we all are eager to see that and see exactly how well it works. […]
Marshall: The side effect report for this drug hasn't been out, but for oral antiviral medications in general, don't they sometimes integrate into the genetic makeup of the virus and cause a lot of mutations to help destroy it?
Schaffner: [Apparently understanding what Marshall really wanted to ask about:] That's been actually already out in the social media and a matter of concern. Believe me, the company was -- the company scientists -- were well aware of this. The Food and Drug Administration scientists were well aware of it. And as the various laboratory and animal studies went forward, they looked into this question very, very carefully. It seems not to be an issue. Otherwise, they would never have permitted trials in human beings to take place.
Schaffner’s answer to whether the drug is dangerous, essentially, is that if a human trial has been approved, the corresponding thing being trialed must have already been proven to be safe. What fantastic news! Why, one wonders why we even bother with trials anymore, including to screen for cancer and fertility harms! - oh, I forgot, we don’t.17
Schaffner’s claim is not only arrogant, but ignorant. Not only do animal trials for Molnupiravir appear to have been limited, focused on infection outcomes only, or with safety results that are seemingly impossible to find18, but nucleoside analog “antiviral” candidates have turned out to pass pre-clinical stages only to reveal toxic effects in human trials before.
This is both intuitive - lab animals do not live as long as humans, and cannot be used to “model” human cancer risks in the first place - and easily revealed by researching the history of this platform. Schaffner, however, is apparently not aware of that history. Neither does anybody else seem to be. In fact the only people who seem to understand just how intrinsically disruptive nucleoside analogs are to host cellular metabolism are the researchers seeking to develop them.19 In a revealing 2018 review of the research by Lu, G. et al., for example (emphasis added):20
There is a growing body of evidence suggesting that some [as in, any which are directly convertible to triphosphate form, including Molnupiravir] ribonucleoside/ribonucleotide analogs may be incorporated into mitochondrial RNA by human mitochondrial DNA-dependent RNA polymerase (POLRMT) and disrupt mitochondrial RNA synthesis. […]
The failure of ribonucleoside/ribonucleotide analogs during clinical development has been due to toxicity that was not detected during preclinical studies.
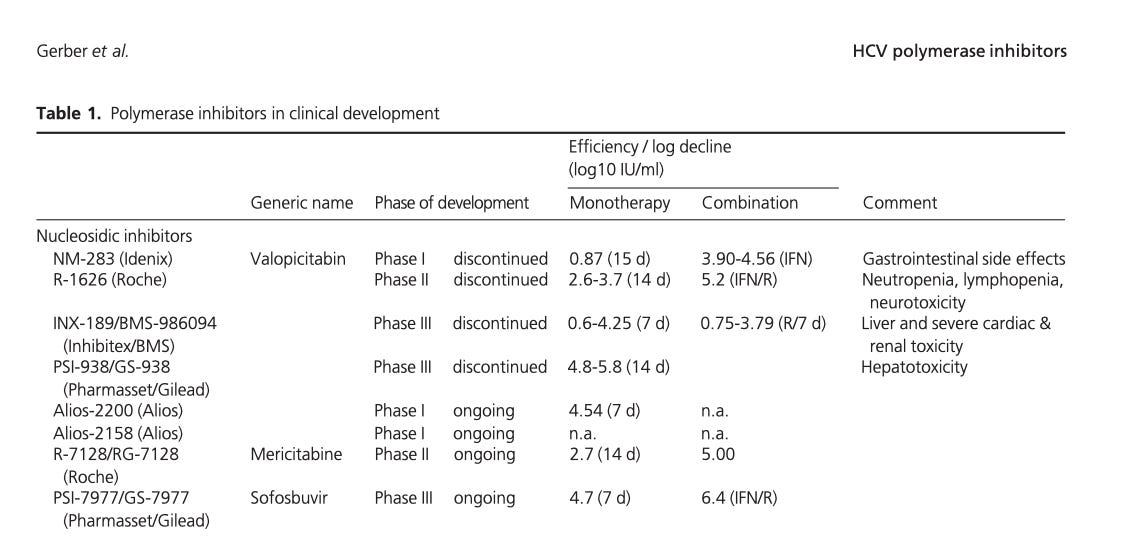
This, in fact, seems only to refer to “Valopicitabin,” which was canceled in four separate trials despite having sailed through pre-clinical screening. From a 2013 review by Gerber, L. et al. (here “nucleosidic inhibitor” refers to nucleoside analogs):21
“Mericitabine” went on to be cancelled for low efficacy - indicating it was only found to be “safe” because it didn’t work very well - and Sofosbuvir is only used in conjunction with Ribavirin (the “R” in the combination trials above), meaning it carries all the same warnings by default, and its own mutagenic and metabolic harms are not evaluable in the real world.
Schaffner’s patronizing, nauseating, idiotic, irresponsible assurance that predatory pharmaceutical companies wouldn’t bring a drug to human trials before ruling out harms, therefor, is easily refuted by the history of the very category Molnupiravir belongs to.
A review from February, 2020, meanwhile, asserts that human trials for Molnupiravir will be preceded by “2 species” dosage studies - not to rule out toxicity, but to calibrate the original dose according to observed harms. Here the authors may only be rephrasing earlier literature from authors associated with DTRA (see below); there is no citation, and once again the actual results of these trials are seemingly impossible to find. This ambiguous suggestion contradicts Schaffner’s sanguine take on affairs. At best, it seems, the animal trials were used to arbitrarily establish the ceiling of danger that current human trial participants are being exposed to (emphasis added):22
Toxicity concerns are a major potential liability of ribonucleoside analogs as underscored, for instance, by the compromised safety profile of [Favipiravir]. A primary source for adverse effects is incorporation of the ribonucleoside analogs into host RNAs by nuclear and mitochondrial RNA polymerases. The integrity of mitochondrial transcripts is particularly at risk due to the proofreading inability of mitochondrial RNA polymerases in contrast to nuclear RNA Pol II. Not surprisingly, [the Molnupiravir “M” package] was incorporated by human mitochondrial polymerases in in vitro studies and an elevated frequency of [“M”]-characteristic transition events was noted after prolonged exposure of primary human airway epithelium cells to the drug. However, the transition mutation frequency in ferret lung tissue was unchanged from that in controls after extended treatment of animals with 200 mg/kg daily dose and thus over 14-times the efficacious dose for influenza therapy [but the drug goes to every tissue, not just the lungs!]. Potential adverse effects of [Molnupiravir] on embryo development are untested at present, and formal 2-species multi-dose toxicity studies will be required to establish initial human doses examined in phase 1 clinical trials.
And of Favipiravir, which is approved for limited use against influenza in Japan, the same review states:
Safety testing revealed that [Favipiravir] is teratogenic and embryotoxic at concentrations close to the approved human dose.
If the entire push to develop nucleoside analogs for antiviral use is starting to seem incredibly crazy, that’s because it is.23
Nucleoside analogs, as I argued in “Doppelgänger,” are not “antivirals.” The mechanism of “viral replication” which they seek to sabotage is intrinsic to cellular metabolism, DNA repair, gene expression, cellular division, and sperm cell longevity. Ribavirin, Molnupiravir, and all other analogs in development, essentially seek to cut off the face to spite the nose. It cannot be done without short and long term harms, and profound risks to fertility. This is not a “therapeutic” platform - it is chemotherapy for the genome. It is an appropriate accident at best, that this particular drug was developed with funding from the US Defense Threat Reduction Agency (Ridgeback and Merck only took over after six years of DTRA-funded research24).
And yet not only are nucleoside analog “antivirals” still being aggressively researched, they reside within a cultural blindspot both within the industry and the public at large - they are lethal bioweapons on the scale of Anthrax, inaccurately imagined as “kind of similar to Tamiflu.”
Is there any chance left that the public won’t have to be sacrificed in droves for the profession to take the obvious toxicity of these drugs seriously?
(jump link anchor)
As the Covid-vaccinated leave the infection efficacy window, they should be adding to the multiplier for new cases. This multiplier should be more than 1 and rising at the beginning of the wave and less than 1 and dropping wherever the wave has peaked. The result should be that the descent curve is less steep than the rising curve; with the potential for an eventual plateau or reversal. This is sort-of visible in some southern states, but not others; and where it is visible it can be attributed to coincidental school openings anyway. A sampling:
Sort of:
And not:
(jump link anchor)
See https://www.gov.uk/government/publications/covid-19-vaccine-weekly-surveillance-reports - Accessed October 15, 2021.
(Update, October 19: The original version of this post did not include a disclaimer about severe outcome efficacy, though in my head I imagined that I must have snuck one in per usual.)
Confirming true “negative efficacy” would require much more rigorous methods, such as sampling the two groups for levels of antibodies to the “N” protein, to see if the Covid-vaccinated really did end up experiencing more infections overall.
Hospitalization rates, per the same report, indicate severe outcome efficacy remains strong - though, triple-dosing has likely begun to distort the data here. For my discussion on “Covid vaccine immune response” which offers my theory for why severe outcome efficacy is durable, see “Boostermania!”:
See https://www.gov.uk/government/statistics/national-flu-and-covid-19-surveillance-reports-2021-to-2022-season - Accessed October 15, 2021.
See https://www.bhf.org.uk/informationsupport/heart-matters-magazine/news/coronavirus-and-your-health/covid-booster-vaccine - Accessed October 15, 2021:
The booster programme started on September 16 and is likely to last at least throughout the autumn.
Boosters will be given in the same order of priority as for the initial vaccine, as follows:
1.Care home residents and staff 2.People aged 80 and over, and frontline health and social care workers […] 6.Adults who are at higher risk from Covid-19, including people with heart and circulatory conditions […] 9.People aged 50 and over
See Shearing, Hazel. “Covid-19: Two million booster jabs given in England.” (2021, October 10.) bbc.com.
(jump link anchor)
See Khazan, Olga. “Is There Another Reason Biden Likes Boosters?” (2021, October 5.) The Atlantic.
I think some proponents of the “worry window” and “vaccine wave” theories used the spring waves as evidence, at the time, that the Covid vaccine rollouts are associated with a surge in cases. I continue to doubt this - and I think the Canada explanation renders many of the previously wielded arguments circular at best.
The Pacific Northwest is an outlier, with both a significant spring wave and a larger autumn wave.
(jump link anchor)
(Emphasis added.) Lu, G. et al. (2018.) “Simple In Vitro Assay To Evaluate the Incorporation Efficiency of Ribonucleotide Analog 5′-Triphosphates into RNA by Human Mitochondrial DNA-Dependent RNA Polymerase.” ASM Journals. Vol. 62, No. 2.
Marshall, Serena. Salahi, Lara. “Merck's COVID Pill Has Been Called a 'Game Changer' -- Is It?” (2021, October 13.) MedPage Today.
See https://www.drugs.com/monograph/ribavirin.html - Accessed October 15, 2021
A 2020 on development of Molnupiravir alludes to pending “28-Day” studies on dogs and rats, without linking or citing any source for results. See Sticher, Z. et al. (2020.) “Analysis of the Potential for N4-Hydroxycytidine To Inhibit Mitochondrial Replication and Function.” Antimicrobial Agents and Chemotherapy. 2020 Feb; 64(2): e01719-19.
“Chronic-toxicity studies (28 days) are under way in rats and dogs, and every effort will be made to monitor for toxicities arising from mitochondrial dysfunction.”
Perhaps drug development wouldn’t be so expensive if companies didn’t focus exclusively on their stupidest ideas.
(Lu, G. et al.)
Gerber, L. et al. (2013.) “New therapeutic strategies in HCV: polymerase inhibitors.” Liver International. Volume 33, Issue 1. Pages 85-92.
Toots M. Plemper, R. (2020.) “Next-generation direct-acting influenza therapeutics.” Translational Research. VOLUME 220, P33-42, JUNE 01, 2020.
Molnupiravir is referred to a EIDD-2801 and Favipiravir as T-705 in the text, hence my substitutions.
Regarding the suggestion that this is only rephrasing previous DTRA literature, the most plausible candidate is the study in footnote 18.
And, as I argued in “Dopplegänger,” likely an accident resulting from the fact that Ribavirin was approved for use over a decade before its mechanism was understood - resulting in a back door precedent for an obviously stupid idea.
See Abinader, Luis Gil. “US government rights in patents on Molnupiravir, based upon funding of R&D at Emory University.” (2021, October 4.) Knowledge Ecology International.

















Hey Brian,
Did you see what's going with the UK S and N antibody testing?
https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1025358/Vaccine-surveillance-report-week-41.pdf
Page 21-23 figures 3, 4a, 4b
Interesting that there is a plateau in Nucleocapsid antibodies given that we'd expect S to rise as more Brits have gotten their gmo-shot(s). Is no one else developing N antibodies, even after recovering from all of the breakthrough infections? Or, is it too soon to tell???
Remember that St. Jude study where the N T-cells seemed to drop off on 6 of 10 patients post jab and we didn't have enough samples to really know what was going on?
Great, so we're going from a not-so-safe vaccine to a not-so-safe drug. Meanwhile, other countries like Thailand and India have already done some legwork on herbal remedies that are safe and possibly effective in mitigating covid...the Western world is so strangely backwards with its excessive focus on new technologies.